Description:
This project involved making art for the new San Marin Science Building. Through this project we learned about the chemistry behind a variety of artistic techniques. My partner and I decided to make a larger scale version of the ornament that we did earlier in class. To create our piece we took a sheet of galvanized iron and reacted it with hydrochloric acid to create pure iron and hydrogen gas. Then, we reacted the exposed iron with cupric nitrate to create iron nitrate and copper in a single replacement reaction. This reaction did not go as smoothly as we anticipated and there were many black marks left behind from the reaction. To alleviate this, we heated the piece under a bunsen burner to create an interesting oxidation reaction.
Formulas of Reactions:
FeZn + HCl -> Fe +ZnCl + H
Fe + Cu(NO3) -> Fe(NO3) + Cu
Fe + O2 -> FeO2
The last reaction needed heat as a catalyst.
Project Reflection:
This project was a substantial learning experience for me. During this project I had some high points and also some low points. One high point during the project was finishing the project despite having a time crunch. One major low point in the project for me was when my partner and I were behind a few days in the work. One C in the graduate profile that I try to practice is being a Conscientious Learner because that skill set is arguably very important for succeeding in a career. Because I did not move quickly enough I had to work harder to catch up; this is a major failure to meet the expectation of a conscientious learner. Next, one thing that I believe I did well in the project was collaboration with my partner. For the most part we were able to work well together and finish everything we had to in time. Next, one more thing I felt needed improvement was my critical thinking skills. I settled on doing a larger scale version of the ornament lab that we did earlier in this unit. I felt that this assignment was not as creative or unique as I would have liked it to be, especially with the potential to do a lot of unique things with this project. However, my partner and I also lacked time, but next time I would like to try to make something more interesting.
Concepts From this Project:
In this project we learned about a variety of chemistry concepts that we integrated in our project.
Chemical Equation:
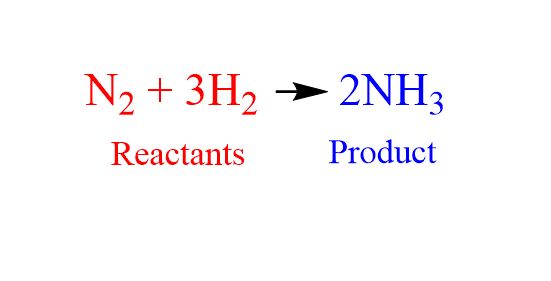
A chemical equation represents a chemical reaction that occurs. One example of an equation would be the one on the left that shows the formation of ammonia through a synthesis reaction. On the left side of the equation is the reactants which are the chemicals that react initially, on the other side are the products which are what is created from the reaction. Instead of an equals sign chemical equations use an arrow to represent that while both sides are equal the reaction may not be easily invertible.
Balancing Equations:
To accurately describe a chemical reaction a chemical equation has to be balanced. The law of conservation of mass applies to chemical equations: both sides must have the same amount of atoms of each individual element. To put equations in their proper form we had to balance the equations.
Valence Shell and Forming Compounds:
In the current model of the atom that scientists have there is an electron cloud that orbits the nucleus of the atom. These electrons orbit at different distances from the atom. The valence shell is the outermost shell of electrons and these valence electrons are a major factor in what compounds form. Elements try to have a complete valence shell of 8 electrons. If they do not have 8 then they have a charge. Certain elements and compounds have a positive or negative charge. If they have a positive charge they are called cations, if they have a negative charge they are called anions.
For instance in the example above the ammonia molecule won't form unless it has a neutral charge. Hydrogen has a charge of +1 and nitrogen has a charge of -3. To form the molecule there are 3 hydrogen atoms so the charges balance and the ammonia has a neutral charge. The number of hydrogen atoms is represented with a subscript.
Types of Reactions:
Many chemical reactions fit general profiles of reactions that we can understand and use to predict how things will occur.
Catalysts:
Reactions happen at certain speeds depending on the chemicals that are involved. However, reactions can often be sped up to be completed faster. A catalyst is something that will do this. For example, heat is often a catalyst for reactions.
Single Displacement Reaction:
A single displacement reaction follows the general form AX + B --> AB + X. As the name goes, the reaction involves a single replacement of one element in a compound. For this reaction to occur the element, X in the compound AX has to be less reactive than B. The chance of a reaction like this occurring depends on the element's place in the activity series.
Double Displacement Reaction:
A double displacement or double replacement reaction follows the general form AX + BY -> AY + BX. In this reaction two compounds Y and X swap places. In the reaction A and B are both cations and X and Y are anions. One example of a double replacement reaction is AgNO3 + NaCl → AgCl + NaNO3. In this reaction the silver and sodium cations swap their nitrate and the chlorine is also swapped.
Decomposition Reaction:
In a decomposition reaction a compound decomposes into multiple parts. They follow a general form AB -> A + B.
2 H2O2 → 2 H2O + O2
Is an example of a decomposition reaction. Hydrogen peroxide decomposes into water and oxygen.
Synthesis Reaction:
In a synthesis reaction a compound is formed by multiple parts. They follow the general form A + B -> AB. One example of a synthesis reaction is the reaction to form water 2H + 02 -> H20. Both molecules synthesize together to create water.
Combustion Reaction:
They follow the general form ... (Hydrocarbon) + 02 -> CO2 + H2O. A hydrocarbon is any compound that involves hydrogen and carbon, one example of this is methane. Here is an example of a combustion reaction. In this reaction methane gas reacts with oxygen to create carbon dioxide and water. The reaction creates heat and energy. -
CH4(g) + 2 O2(g) → CO2(g) + 2 H2O(g)
